Benefits
- Monitor chimerism changes in time
- Generate lab protocols based on your input
- RUO & CE-IVD
Description
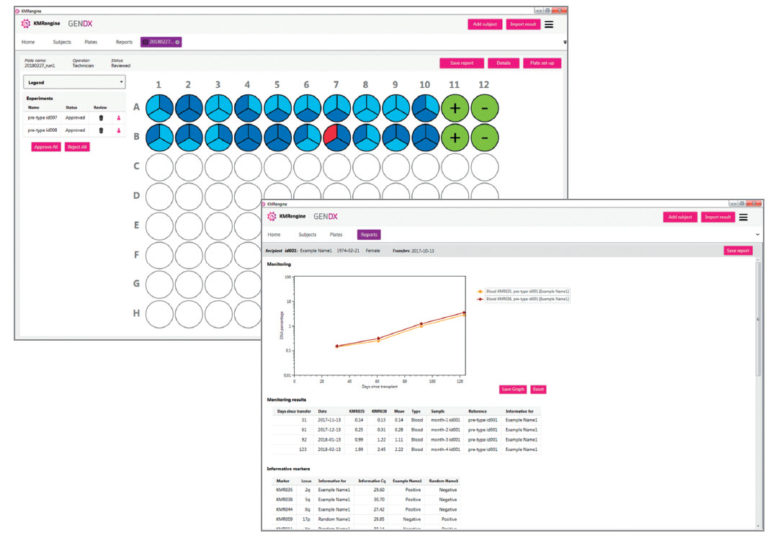
KMRengine is our highly intuitive software for the chimerism monitoring workflow. KMRengine guides you through the entire workflow, from genotyping to monitoring, from the first time point to the most recent time point of monitoring. Available as RUO and CE-marked IVD.
You have questions or want to arrange a demo?
See specifications