Human Leukocyte Antigen (HLA) proteins have long been central in the transplantation field, because of their critical role on transplant patient outcomes and the survival of transplanted organs and cells. Over the past decades, HLA proteins have gained increasing attention as a critical biomarker in the Precision Medicine space. This has been particularly for development of immuno-oncology treatments, evidenced by the number of HLA-restricted therapies entering clinical trials that continues to grow rapidly (see figure 1). Because these therapies are designed to work only in patients carrying specific HLA types, accurately identifying eligible patients through HLA testing is becoming an increasingly important step in bringing these treatments to the clinic.
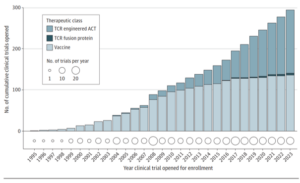
Figure 1: The annual cumulative number of clinical trials worldwide investigating HLA-restricted therapeutics for any malignant indication. Over the past two decades the number of trials with HLA-restricted therapies has clearly increased. Colour shading of each bar indicates the relative contribution from each HLA-restricted therapeutic class, T-cell receptor (TCR) engineered adoptive cell therapy (ACT), TCR fusion protein and HLA-restricted vaccine. Adapted from Gormally, M. V., et al. (2025) [1].
In 2022, tebentafusp was the first HLA-restricted therapy that received approval from the Food and Drug Administration (FDA), for the use in unresectable or metastatic uveal melanoma in HLA-A*02:01 positive adults. In 2024, another HLA-restricted therapy, afamitresgene autoleucel, was approved by the FDA for the treatment of unresectable or metastatic synovial sarcoma in patients positive for HLA-A*02:01P, HLA-A*02:02P, HLA-A*02:03P, or HLA-A*02:06P. Given the rapid increase in HLA-restricted therapies entering clinical trials, it is likely that many more of these treatments will reach patients in the coming years.
Why use HLA proteins or HLA-restriction in new therapies?
To understand the increasing focus on HLA-restricted therapies, appreciating the role of HLA is of importance. HLA proteins are an important step in initiating adaptive immune responses. HLA proteins help the immune system distinguish “self” from “non-self” by presenting short peptide fragments (antigens) to T cells. This antigen presentation is a key step in enabling targeted T-cell immune responses. The antigens presented by HLA molecules can originate from the body’s own proteins or from pathogens such as viruses and bacteria. Differences in HLA molecules between donor and recipient are also a key reason why immune responses can occur after transplantation. Following presentation of antigens through HLA proteins, T-cells can recognize the complex of an HLA protein loaded with a peptide through a highly specific T-cell receptor (TCR) and, if the right signals are there, trigger T-cell activation. T-cell activation can subsequently initiate a very specific immune response to the recognized threat. This key role of HLA in T-cell activation makes HLA-restricted antigen presentation a very interesting focus for immunotherapies.
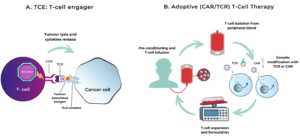
The HLA-restriction of the two therapies, tebentafusp and afamitresgene autoleucel, are based on different mechanisms (see figure 2). Tebentafusp is an example of a so-called T-cell engager (TCE) [2]. It acts by bringing patient T-cells in close proximity with melanoma cells, in order to allow the T-cells to more effectively kill the cancerous cells. This is achieved by the binding of tebentafusp to a protein on patient T-cells (CD3) and to a protein commonly present on melanoma cells (gp100) presented by a HLA-A*02:01 protein.
Afamitresgene autoleucel on the other hand is an example of an adoptive cell therapy (ACT) based on T-cell receptor (TCR) gene therapy and uses the patient’s own T-cells [3]. Adoptive cell therapy (ACT) is a form of immunotherapy that modifies and infuses a patient’s own immune cells to destroy cancer. Most commonly known strategies for ACT are chimeric antigen receptor (CAR) T-cell therapy (often HLA independent), TCR-engineered T-cells (often HLA-restricted). In the case of afamitresgene autoleucel, the patient’s own T-cells are collected, genetically engineered in a lab to express a specific TCR. This TCR specifically recognizes a protein commonly found in synovial sarcoma cells (MAGE-A4) presented by specific HLA molecules, HLA-A*02:01P, HLA-A*02:02P, HLA-A*02:03P, or HLA-A*02:06P. Subsequently the engineered T-cells are expanded and re-infused into the patient.
Figure 2: Schematic representation of two HLA-restricted therapy strategies.
- Visualization of a T-cell engager (TCE). A TCE is a bispecific fusion protein that operates by physically linking a patient’s T-cells to tumour cells carrying a specific marker. By bringing the T-cells into very close contact with the cancer cells and activating them through CD3, it triggers a focused immune attack that kills the tumour cells. Adapted from Rallis et al., 2021 [4].
- Visualization of Adoptive Cell Therapy (ACT). T-cells are isolated from peripheral blood from a patient. Subsequently, specific T-cells are genetically modified with an engineered T-cell receptor (TCR) or chimeric antigen receptor (CAR), designed to specifically recognize a tumour antigen. Following successful modification, the modified T-cells are expanded in the lab and finally infused into the patient. The engineered T cells can now recognize the target antigen on tumour cells and trigger a targeted immune response. Adapted from Met et al., 2019 [5].
Beyond these two approved therapies there are multiple examples of therapies that leverage HLA and are currently being tested in late stage clinical trials. These therapies include but not limited to HLA-restricted adoptive cell therapies, HLA-restricted epitope vaccines and several different T-cell engagers. Examples of such trials are:
- The ARTEMIA clinical trial [6], investigating safety and efficacy of HLA-A*02 restricted cancer vaccine (OSE2101) in non-small cell lung carcinoma patients;
- the FLAMINGO-01 trial [7], investigating safety and efficacy of a HER2/neu peptide in combination with GM-CSF (GLSI-100) in HLA-A*02 positive breast cancer patients;
- the SUPRAME trial [8], investigating safety and efficacy of TCR-T ACT targeting PRAME (ACTengine IMA203) in HLA-A*02:01 positive patients with unresectable or metastatic cutaneous melanoma;
- and the PRISM-MEL-301 trial [9], investigating safety and efficacy of a TCE targeting CD3 and PRAME (IMC-F106C) in HLA-A*02:01-positive participants with previously untreated advanced melanoma.
Taken together, these examples highlight how immunotherapies are increasingly designed to harness the central role that HLA proteins play in the initiation of specific T-cell responses. However, the same HLA-restriction that enables these therapies to precisely target tumour cells also means that only patients carrying specific HLA alleles can benefit from these therapies. This introduces an important practical question: how do clinicians know which patients are eligible for these therapies?
This is where companion diagnostics (CDx) play a central role. HLA typing assays used as companion diagnostics allow clinicians to screen patients and determine whether they are likely to benefit from a specific HLA-restricted therapy.
As the number of HLA-targeted therapies continues to grow, robust and accurate HLA testing will become increasingly important to identify eligible patients and ensure that those patients who are most likely to benefit, can gain access to these treatments.
Interested in learning more?
Do you want to know more about HLA-restricted therapies, the importance of HLA in the Precision Medicine field or the impact of HLA on therapy success? Stay tuned for the next blog post.
Do you want to learn more about our Companion Diagnostics services and how we can be your partner from start to finish? Read more on our dedicated Companion Diagnostics webpage or get in touch with our team.
Authors
Jules Petit – Project manager CDx

Jules holds a PhD in Molecular Immunology. After obtaining his PhD, Jules worked in different positions, both in an academic and in a pharmacological industry setting, before starting at GenDx. At GenDx he manages the different Companion Diagnostics programs and supports the scientific, strategic and regulatory integration of these projects.
Valentina Manzini – Project Coordinator CDx

Valentina holds a PhD in Molecular Biology, with a background in tumor biology. She is currently working at GenDx, supporting the Companion Diagnostics team by ensuring projects run smoothly, performing regulatory submissions and coordinating IVD activities of clinical trials involving both drugs and CDx.
Relevant references
[1] Gormally, M. V., Chen, M. F., Noronha, A. M., Panageas, K., Reynolds, M., Kohlasch, K., … & Donoghue, M. T. (2025). Next-generation sequencing for HLA genotype screening and matching to HLA-restricted therapies. JAMA oncology, 11(1), 74-76.
[2] https://www.cancer.gov/about-cancer/treatment/drugs/tebentafusp-tebn
[3] https://www.cancer.gov/about-cancer/treatment/drugs/afamitresgene-autoleucel
[4] T-cell-based Immunotherapies for Haematological Cancers, Part A: A SWOT Analysis of Immune Checkpoint Inhibitors (ICIs) and Bispecific T-Cell Engagers (BiTEs)
[5] Met, Ö., Jensen, K. M., Chamberlain, C. A., Donia, M., & Svane, I. M. (2019, January). Principles of adoptive T cell therapy in cancer. In Seminars in immunopathology (Vol. 41, No. 1, pp. 49-58). Berlin/Heidelberg: Springer Berlin Heidelberg.
[6] Liu, S. V., Dziadziuszko, R., Viteri, S., Cappuzzo, F., Comis, S., Gabarre, V., … & Besse, B. (2024). 1397TiP Phase III trial of the therapeutic cancer vaccine OSE2101 versus docetaxel in patients with metastatic non-small cell lung cancer and secondary resistance to immunotherapy. Annals of Oncology, 35, S874-S875.
[7] Patel, S., Thompson, J., Patel, M., Daugherty, F. J., & Rimawi, M. F. (2023). Phase III study to evaluate the efficacy and safety of GLSI-100 (GP2+ GM-CSF) in breast cancer patients with residual disease or high-risk PCR after both neoadjuvant and postoperative adjuvant anti-HER2 therapy: Flamingo-01.
[8] Luke, J. J., Betof Warner, A., Chmielowski, B., Diab, A., Gebhardt, C., Hernandez-Aya, L. F., … & Britten, C. (2025). SUPRAME: A phase 3 trial comparing IMA203, an engineered T-cell receptor expressing T cell therapy (TCR-T) vs investigator’s choice in patients with previously treated advanced cutaneous melanoma.
[9] Long, G. V., Atkinson, V., Ascierto, P. A., Davar, D., Hamid, O., Robert, C., … & Schadendorf, D. (2024). A phase 3 trial of IMC-F106C (PRAME x CD3) plus nivolumab versus standard nivolumab regimens in HLA-A* 02: 01+ patients with previously untreated advanced melanoma (PRISM-MEL-301).